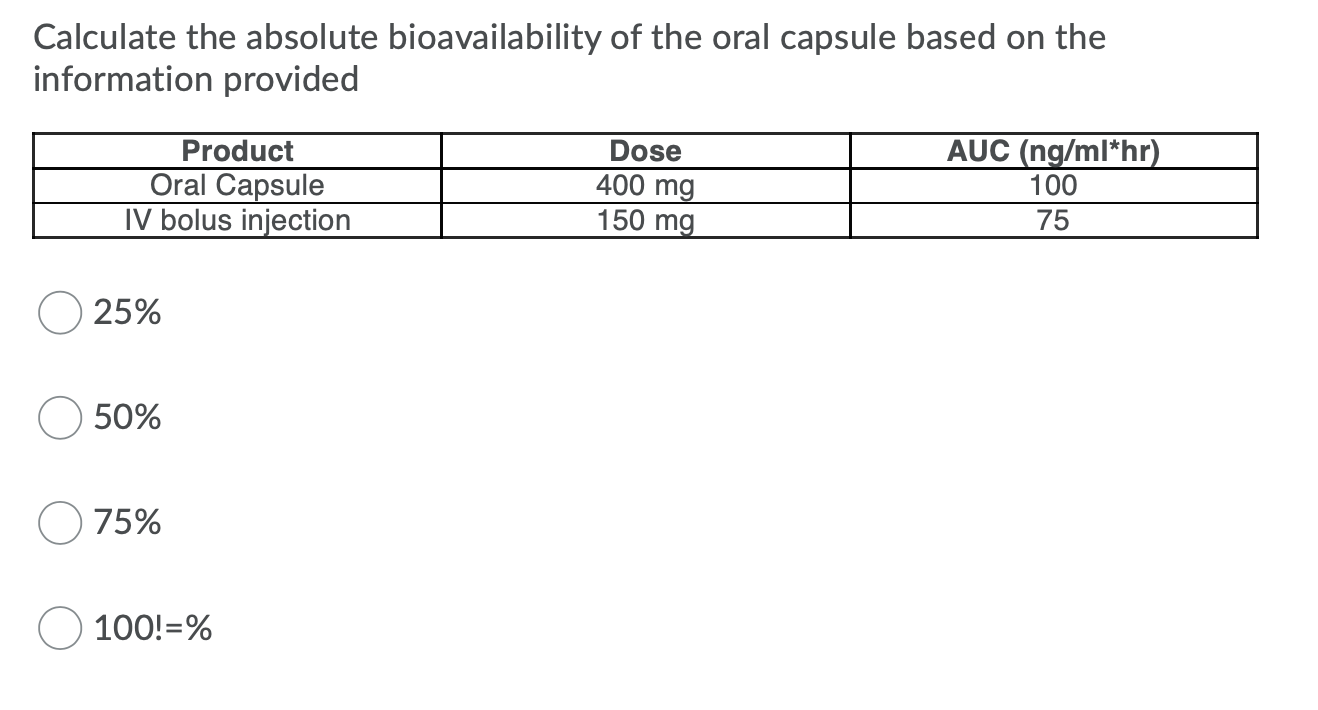
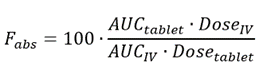Stable Isotopically Labeled Intravenous Microdose Pharmacokinetic Trials as a Tool to Assess Absolute Bioavailability: Feasibility and Paradigm to Apply for Protein Kinase Inhibitors in Oncology - Roosendaal - 2020 - Clinical Pharmacology
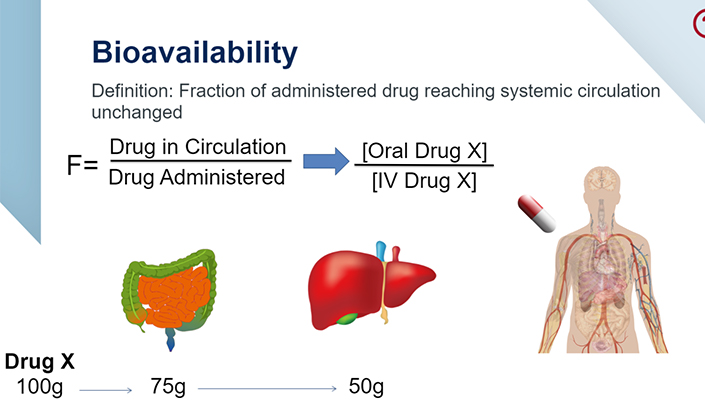
Drug Dosing Calculations: an interactive approach to maintenance and loading dose calculations | E-Gallery | University of Nebraska Medical Center

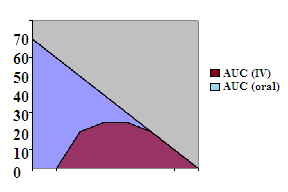
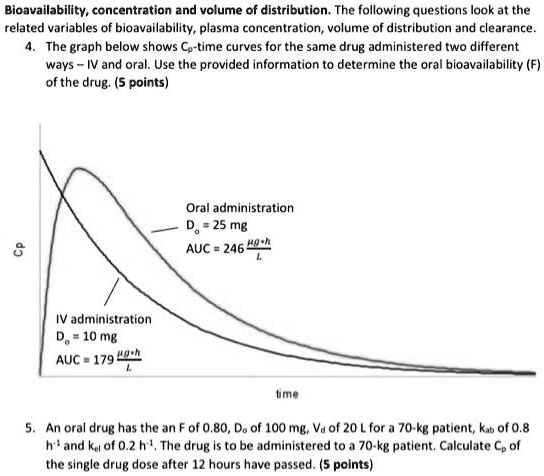
Absolute bioavailability estimated by calculating the AUC ratio between... | Download Scientific Diagram

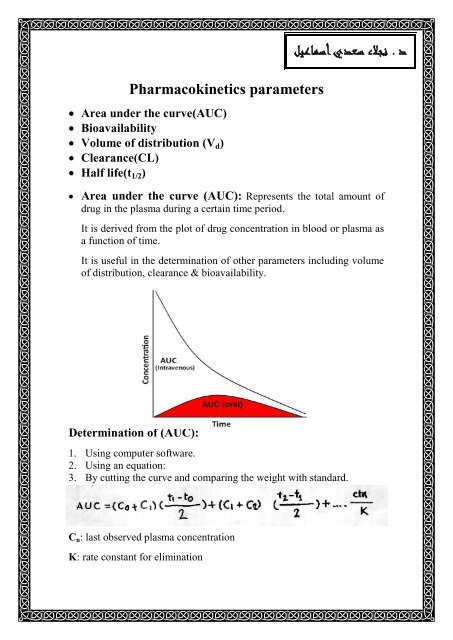

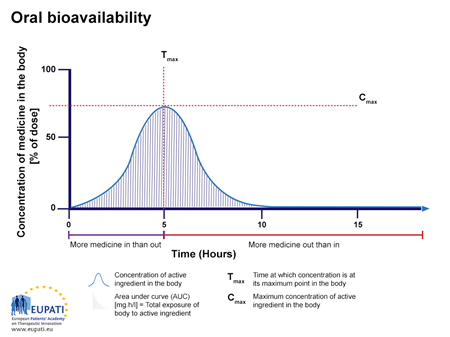

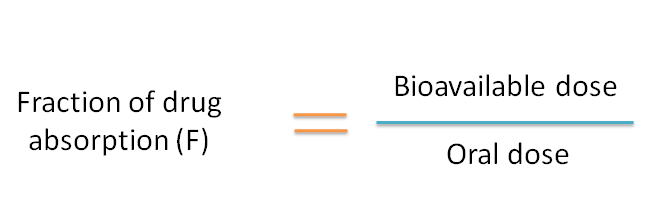
![introduction_to_pharmacokinetics [TUSOM | Pharmwiki] introduction_to_pharmacokinetics [TUSOM | Pharmwiki]](https://tmedweb.tulane.edu/pharmwiki/lib/exe/fetch.php/pk-ba2.png)